AEFI Reporting Application for Medical Colleges
Download MarkDownThe Challenge
The Ministry of Health and Family Welfare (MoHFW), with technical assistance from JSIPL through the Immunization Technical Support Unit (ITSU), needed to strengthen the reporting of Adverse Events Following Immunization (AEFI) from select medical colleges across India. The existing surveillance system had significant gaps in capturing adverse event data from healthcare institutions, which is critical for maintaining vaccine safety profiles and identifying emerging vaccine safety issues.
Key challenges included:
- Fragmented Reporting: Doctors and faculty members across approximately 40 medical colleges in 8 cities (Lucknow, Bangalore, Bhopal, Jaipur, Bhubaneswar, Pune, Hyderabad, Chennai) had no unified digital mechanism to report AEFI cases
- Connectivity Constraints: Many medical college settings had intermittent internet connectivity, making real-time data submission unreliable
- Complex Data Capture: The Case Reporting Form (CRF) required capturing detailed clinical information including patient identifiers, treatment details, case diagnosis, vaccination history, and supporting investigation records, along with scanned hospital record images
- Multi-Level Access Requirements: Four distinct user roles (Notifier, Nodal Officer, State Manager, National Admin) each required different access levels and permissions
- Automated Notification Needs: Upon CRF submission, automatic notifications needed to reach multiple stakeholders — District Immunization Officer (DIO), SEPIO, SMO (WHO), SPM, ITSU, and the reporter — via email with attached PDF reports
Velocity’s Solution
Scope of Work
Velocity developed a comprehensive cross-platform application (web-based, Android, and iOS) for AEFI reporting, enabling medical college staff to capture, submit, and track adverse event cases through an intuitive digital platform with offline capability and automated notification workflows.
Key Features & Deliverables
Cross-Platform Application: Native development for Windows, Android, and iOS platforms with responsive layouts adapting to tablets, desktops, and laptops
Offline-First Architecture: Complete offline data capture with automatic synchronization to the central server upon connectivity restoration
Case Reporting Module: Comprehensive CRF data entry with patient identifiers, treatment details, case diagnosis, vaccination history, and supporting investigation records
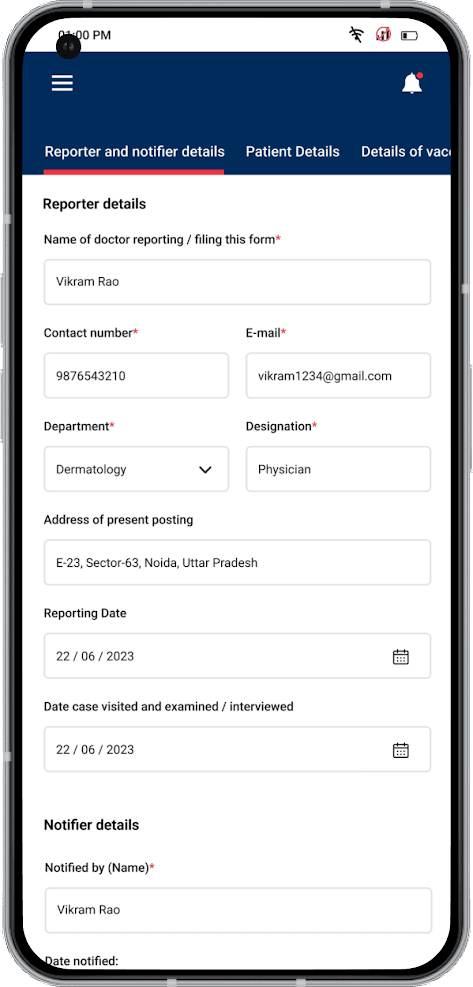
Image Capture and Management: In-app photo/image capture with optimization (approximately 3 MB per image), coded and linked to unique case IDs, with standard image customization functions
Automated PDF Generation: System generates PDF documents of submitted cases including all captured photos, images, and documents, automatically emailed to DIO, SEPIO, SMO, SPM, ITSU, and the reporter
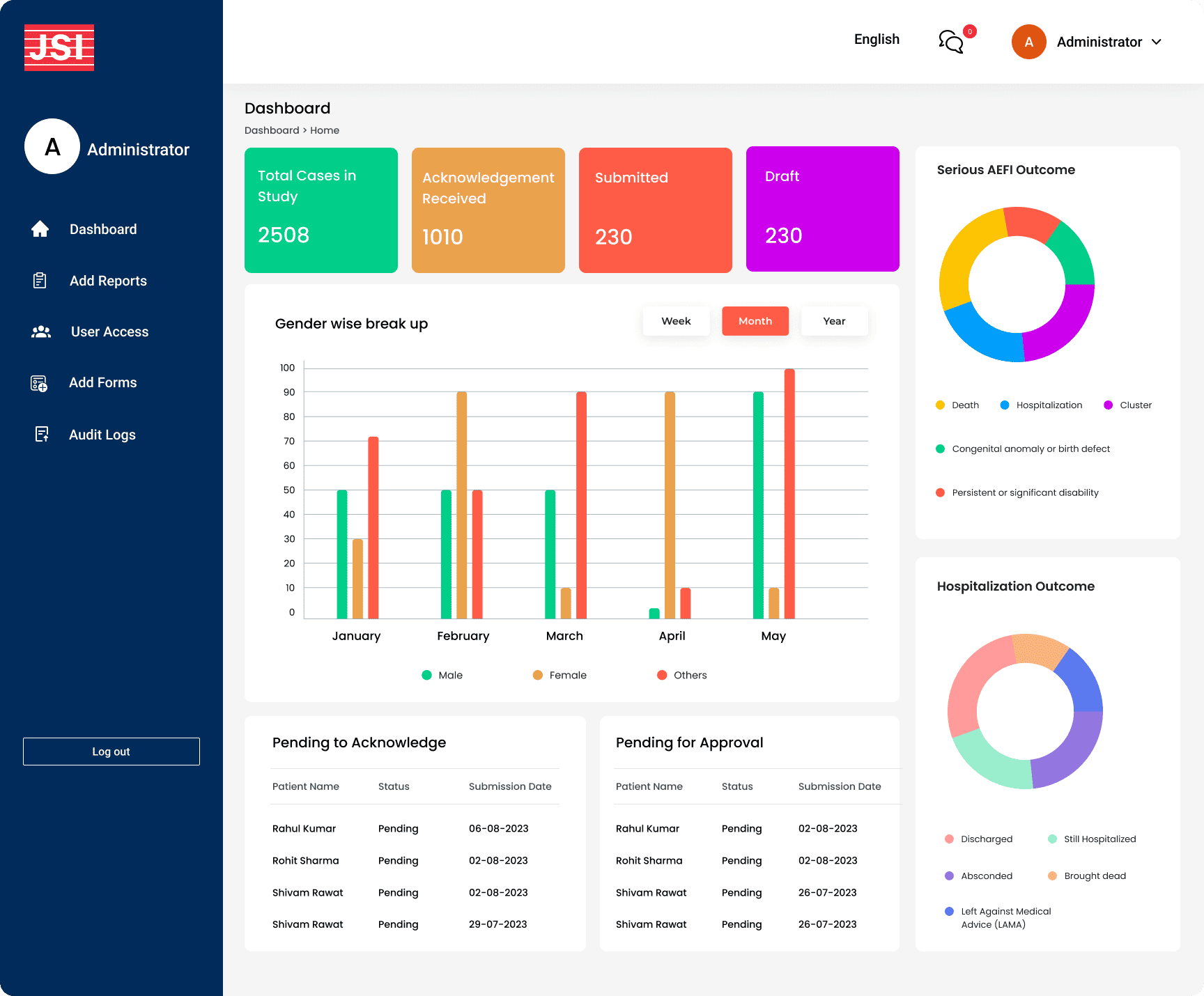
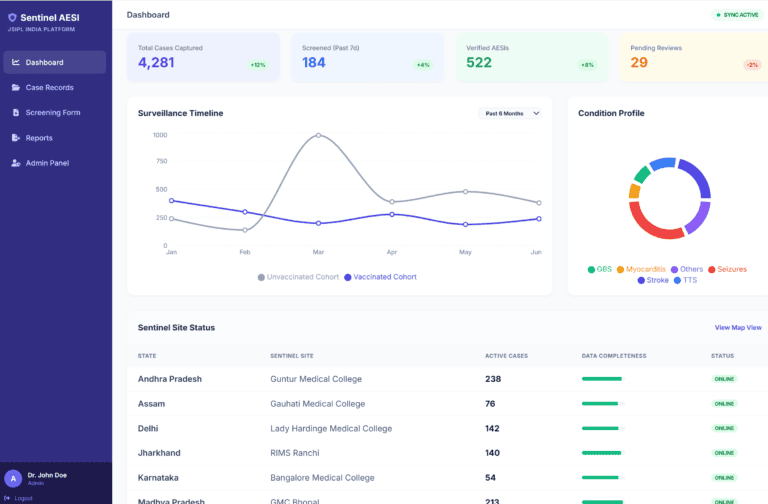
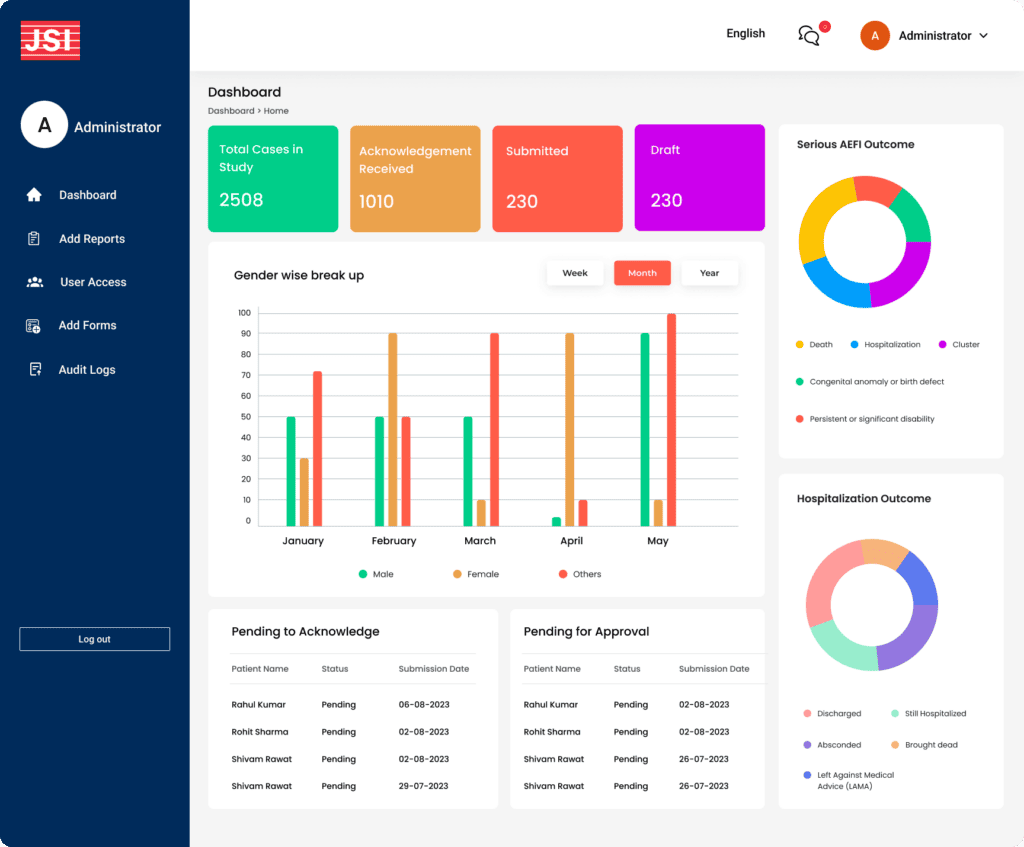
Multi-Level Dashboard System: Role-based dashboards at National, State, Facility, and Notifier levels with summary tables, KPIs, geographical case representation, and demographic breakdowns (age, gender, timeliness, completeness)
User Management Module: Four-tier user rights system (Notifier, Nodal Officer, State Manager, National Admin) with appropriate view/edit/delete permissions
Admin Module: Centralized admin panel with national-level controlling rights for all medical colleges, user verification, and database management
Push Notifications and Alerts: Support for push notifications, alerts, and flash messages to keep users informed of case status updates
Custom Reporting: Ability to generate custom view/download reports with advanced filtering, drill-down functionality, and database search capabilities
- Data Connectivity: Integration with multiple data sources including Power BI, APIs, web links, and Excel spreadsheets with secure API authentication
- Email Authentication: Registered users authenticated via email-based verification system
- Security Audit: CERT-In empaneled security audit completed within four weeks of development
Technology Stack
- Frontend: MS .NET Framework, PHP, React Native, JavaScript
- Backend: MS SQL Server, MongoDB
- Mobile: Native Android SDK, iOS Development Kit
- Email: MTA (Mail Transfer Agent), MUA (Mail User Agent) technology
- Styling: Cascading Style Sheets (CSS) with page-tree style navigation
- Server: Windows Server (HDD 4TB, Cloud Backup/NAS Drive 12TB, RAM 16GB, CPU 4-core)
- Security: CERT-In empaneled audit, encrypted personal data, secure APIs
Implementation Approach
The project followed a rapid, module-based development methodology delivered in 8 weeks:
Week 1 – Inception and Planning
– Inception report with methodology, tools, and activity Gantt chart
– System Requirement Specification (SRS) document
– Responsibility matrix finalization
Week 2 – Core Module Development
– Module 1: User Registration and Login with Single Sign-On authentication
– Module 2: Forms Creation with visual design, screen development, logical configuration
– Test scripts and bug fixing for module and system testing
Week 3 – Analytics and Infrastructure
– Module 3: Dashboard development and custom report generation
– Server configuration and deployment setup
Week 4 – Deployment and Testing
– Production environment setup with user definitions at Medical College, State, and District levels
– User Acceptance Testing (UAT) and Performance Testing
– User manual, troubleshooting manual, administrative manual, and training manual delivery
Weeks 4-8 – Go-Live and Stabilization
– Application deployment on Google Play Store
– Security audit by CERT-In empaneled agency
– Bug fixing and performance optimization
Post-Launch – Maintenance (through March 2024)
– 8x7x300 emergency support (8 hours per day, 7 days a week, approximately 300 days)
– Error resolution, patch deployment, updates, and upgrades
– Dedicated resource person for day-to-day operations
Key Outcomes & Impact
- Nationwide AEFI Coverage: Successfully deployed across approximately 40 medical colleges in 8 major cities, creating India’s first dedicated digital AEFI reporting system for medical colleges
- Real-Time Surveillance: Enabled real-time adverse event reporting and monitoring, contributing to vaccine safety profiles of Routine Immunization vaccines
- Automated Workflow: Eliminated manual notification processes through automated PDF generation and email distribution to all relevant stakeholders
- Offline Resilience: Ensured uninterrupted data capture even in connectivity-challenged medical college environments
- Multi-Level Visibility: Provided national, state, and facility-level dashboards enabling decision-makers to identify emerging vaccine safety issues promptly
- Data-Driven Decision Making: Geographic and demographic visualizations enabled targeted interventions based on AEFI patterns
Why Velocity
Velocity was the ideal partner for this mission-critical public health project based on:
- Healthcare Software Expertise: Proven experience building health information systems for government programs with complex clinical data capture requirements
- Cross-Platform Development: Demonstrated capability in developing native applications across Windows, Android, and iOS platforms
- Offline-First Design: Deep understanding of connectivity challenges in Indian healthcare settings and expertise in building robust offline synchronization mechanisms
- Rapid Delivery: Ability to deliver a fully functional, tested, and deployed application within an aggressive 8-week development timeline
- Government Compliance: Familiarity with Government of India IT standards, MoHFW metadata standards, and CERT-In security audit requirements
- Ongoing Support Commitment: Dedicated support model ensuring application reliability throughout the project lifecycle